Dynamic Properties of Amino Acid Based Surfactants
Personal hygiene regimes have become an important part of our everyday lives and this presents new challenges for the formulation of personal care products. Consumers increasingly demand not only excellent cleansing performance but also a continuing pleasing effect after rinseoff application.
Furthermore, the products must meet the expectations created by increasing awareness of environmental concerns and be as near to natural as possible. The mildness of the formulations is essential and amino acid-based surfactants such as the salts of cocoyl glutamate and lauroyl sarcosinate have an increasingly important role to play in this developing market (Figure 1).
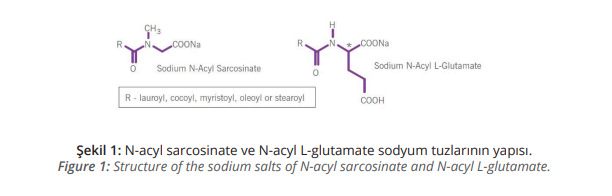
Both substances consist of the joining of a naturally occurring amino acid as a hydrophilic head to a long chain fatty acid as the hydrophobic tail. The ionic structure of the
amino acid part has a high affinity to proteinaceous surfaces such as skin and hair. Studies have shown that these amino acid-based surfactants form a protective monolayer on the skin and this prevents drying out of the skin. This protective monolayer also keeps more aggressive primary or secondary surfactants that may be present away from
the surface of the skin 1.
In stark contrast to standard surfactants such as SLS, SLES and cocamidopropyl betaine (CAPB), the N-acyl sarco[1]sinates and N-acyl glutamates show a capability to buf[1]fer the pH of formulations in the area of 5.5. This buffer capacity allows the preparation of stable formulations without any negative effects on the healthy condition of the skin 4 .
N-acyl glutamates such as Perlastan SC 25 NKW are well tolerated by the skin due to its low tendency to cause skin irritation (hypoallergenic) and absence of pore blocking (non-comedogenic) properties. The transepidermal water loss (TEWL) is a measure for the barrier stability of the epidermis and therefore an indicator for the irritant potential of substances.
In a clinical study the TEWL of human skin was investigated after application of two rinseoff formulations based upon sodium cocoyl glutamate and sodium lauroyl sarcosinate as main components. No significant change in skin moisture of the subjects could be determined, which is to be interpreted as a positive result. Furthermore, the tested rinse-off formulations showed a high degree of acceptance by the subjects in terms of their mild, nourishing, cleansing and foaming effect.
Glutamic acid as a starting material has the advantage that production is carried out by natural fermentation processes. Thus, Perlastan glutamates such as SC 25 NKW, SL and SCG 50 ZPF fulfil the requirements for obtaining certifica[1]tion by various organisations such as COSMOS, ECOCERT, NATRUE, ECO-Label and Nordic Swan.
They can be readily used as the main components or secondary surfactants in formulations when certification as natural cosmetics is required. Their mild and particularly skin-friendly character is combined with a pronounced foaming behaviour and excellent aerobic and anaerobic biodegradability6 .
These positive features make the range of N-acyl sarcosinates and N-acyl glutamates surfactants of choice to meet increasingly sophisticated needs of consumers with respect to naturalness, health and performance.
In this article, the performance of amino acid-based surfactants and their formulations was examined by various methods with regard to their cleansing power, foam ability and foam structure and compared with conventional standard surfactants.
Cleansing Performance
To evaluate cleansing performance of surfactants, the determination of critical micelle concentration (CMC) can be measured by means of the ring tensiometer. The maximum reduction of surface tension of an aqueous surfactant solution is measured as a function of surfactant concentration. The lower the CMC value, the better the surfactant performance. Measurements with the ring tensiometer method have shown that standard anionic surfactants SLS and milder SLES have a CMC value of 1.2 g/l (SLS) and 0.3 g/l (SLES) respectively (Figure 2).
The relatively high value for SLS corresponds to a poor cleansing performance that can only be compensated with a higher level of use of surfactant in the formulation.
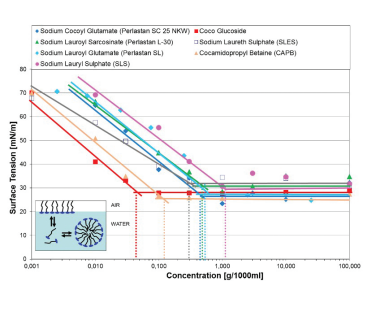
Figure 2: Static surface tension as a function of concentration. CMC of sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium lauroyl glutamate (Perlastan SL), sodium lauroyl sarcosinate (Perlastan L-30) and other common surfactants.
For this reason, it is common practice to include additional surfactant(s) with particularly low CMC in a formulation to reduce the CMC of the formulation as a whole.
A widely used and convenient formulation of cleansing products is the mixture of anionic surfactant SLES and amphoteric CAPB (CMC 0.12g/l). However, the strong ionic
structure of the sulphate group in SLES increases the potential for creating conditions that can show as skin irritation and can lead to intolerance especially if someone has
sensitive skin.
In this case the use of amino acid-based surfactants such as sodium cocoyl glutamate (CMC 0.4 g/l), sodium lauroyl glutamate (CMC 0.4 g/l) or sodium lauroyl
sarcosinate (CMC 0.4 g/l) can reduce the irritant effects and produce a milder formulation.
They may be added at very low levels for instance to SLES and amphoteric surfactants (betaines) formulations to produce significantly milder effects with positive influence on the health of the skin’s surface. The protective monolayer reduces adsorption of aggressive surfactants on the skin and at the same time loss of skin moisture is prevented.
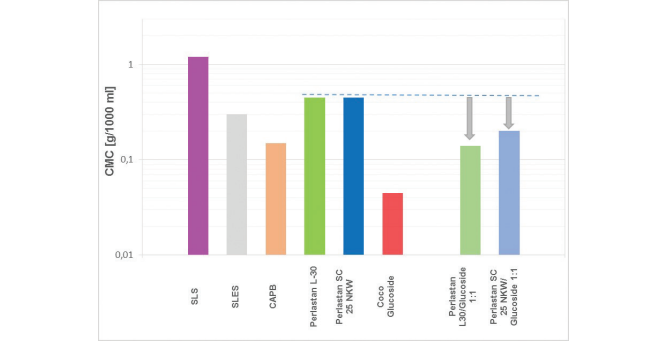
Figure 3: Reduction of the critical micelle concentration (CMC) of sodium cocoyl glutamate (PerlastanSC 25 NKW) and sodium lauroyl sarcosinate (Perlastan L-30) by equivalent combination with coco glucoside (1:1).
When the surfactants are mixed with non-ionic surfactant coco glucoside (CMC 0.045 g/l) in equivalent ratio, the total-CMC is reduced from 0.4 g/l to 0.12g/l or 0.2 g/l (Fig 3). This means that a lower concentration of surfactant is needed to obtain same cleansing performance with excellent foam ability and mildness of the product.
Foaming Behaviour
For a consumer, one of the most important visible indicators of a good cleansing performance is a good foam formation. Rich foaming is associated with good cleansing
ability while low foam is not.
Good foaming also demonstrates visibly that the product is available to spread over the skin. To investigate foam characteristics of Perlastan surfactants a manual RossMiles test was carried out (Figure 4).
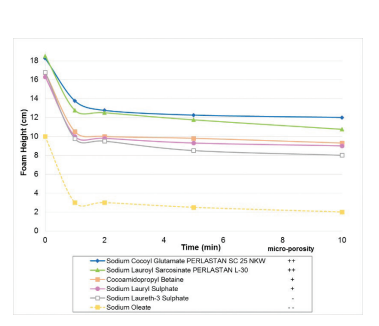
Figure 4: Ross-Miles test with sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium lauroylsarcosinate (Perlastan L-30) and other surfactants (active content of 3 g/l).
This test was performed to determine foam ability and foam stability of surfactant solutions with an active content of 3 g/l (above the CMC). A defined volume of foam was generated by a foam dispenser and the foam height was measured as a function of time. The developed foam height of Perlastan surfactants is in a general equivalent to high foaming standard surfactants such as sulphates and betaines.
It is a positive additional effect that these surfactants form foams that are extremely stable and microporous in structure. In an additional test where defined foam was formed by means of a foam dispenser, the amount of liquid draining from the foams was measured on a time dependent basis.
This is an indication of the amount of liquid in the foam. The results show that sarcosinates and more particularly glutamates provide a stable foam with a high water content that is stable over a longer period (Figure 5).
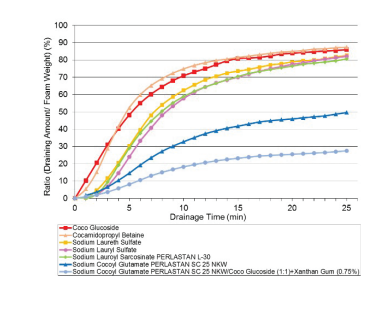
Figure 5: Foam Stability: Time-dependent drainage amount of sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium cocoyl glutamate/coco glucoside/xanthan gum, sodium lauroyl sarcosinate (Perlastan L-30) and other surfactants (active content of 3 g/l).
CAPB and coco glucoside provided the fastest water loss and thereby may be described as being the most unstable and driest foam. A wet foam consistency as demonstrated by amino acid-based surfactants allows a particularly easy application of the product on skin and leaves a pleasant skin-feel.
It was also demonstrated that the relatively unstable foam of a 0.3% coco glucoside solution (active content of 3 g/l) was noticeably stabilised by an equivalent amount of sodium cocoyl glutamate (1:1) and xanthan gum (1%) and gave the best stability under these test conditions.
Conclusion
Amino acid based surfactants provide a lot of positive characteristics, which are of great advantage for formulations of sulphate-free cleansing products. They show pronounced foaming behaviour with particularly high moisture content foam. This makes the products very attractive for rinse-off applications.
In formulations, more aggressive surfactants such as SLS or SLES can be combined with Perlastan surfactants. The irritation potential of the main surfactant is greatly reduced and the resulting formulation benefits from much milder properties. In addition, buffer capacity of glutamate and sarcosinate surfactants in the range between pH 5.0 and pH 6.5 keeps the healthy condition of the skin intact. Furthermore, cleansing power of amino acid-based surfactants can be optimised through the combination with other surfactants which offer a lower CMC.
Due to the excellent aerobic and anaerobic biodegradability Perlastan surfactants are classified as environmentally friendly. Sodium acyl glutamates from products such as Perlastan SC 25 NKW, Perlastan SC 25 NKPF, Perlastan SCG 50 ZPF and Perlastan SL meet different criteria of a wide range of certification processes and can be easily applied in natural cosmetics formulations.
References
1. Sugar M, Schmukker R. Reduzierung der Hautadsorption von Sodium Laureth Sulfat: Ein neuer Weg, die Hautfeuchtigkeit nach Anwendung von Duschprodukten zu erhöhen, SOFW Journal 2001; 127: 3-5.
2. Kanari M, Kawasaki Y, Sakamoto K. J. Soc. Cosm. Chem. 1993; 27: 498.
3. Nnanna IA, Xia J. Protein-Based Surfactants. Surfactant Science Series 2001; 101: 261-270.
4. Husmann M, Weisse J, Wragg P, Wasko J. Perlastan Surfactants Derived from Naturally occurring Amino Acids. Cosmetic Science Technology 2008; 194-201.


 Both substances consist of the joining of a naturally occurring amino acid as a hydrophilic head to a long chain fatty acid as the hydrophobic tail. The ionic structure of the
amino acid part has a high affinity to proteinaceous surfaces such as skin and hair. Studies have shown that these amino acid-based surfactants form a protective monolayer on the skin and this prevents drying out of the skin. This protective monolayer also keeps more aggressive primary or secondary surfactants that may be present away from
the surface of the skin 1.
In stark contrast to standard surfactants such as SLS, SLES and cocamidopropyl betaine (CAPB), the N-acyl sarco[1]sinates and N-acyl glutamates show a capability to buf[1]fer the pH of formulations in the area of 5.5. This buffer capacity allows the preparation of stable formulations without any negative effects on the healthy condition of the skin 4 .
N-acyl glutamates such as Perlastan SC 25 NKW are well tolerated by the skin due to its low tendency to cause skin irritation (hypoallergenic) and absence of pore blocking (non-comedogenic) properties. The transepidermal water loss (TEWL) is a measure for the barrier stability of the epidermis and therefore an indicator for the irritant potential of substances.
In a clinical study the TEWL of human skin was investigated after application of two rinseoff formulations based upon sodium cocoyl glutamate and sodium lauroyl sarcosinate as main components. No significant change in skin moisture of the subjects could be determined, which is to be interpreted as a positive result. Furthermore, the tested rinse-off formulations showed a high degree of acceptance by the subjects in terms of their mild, nourishing, cleansing and foaming effect.
Glutamic acid as a starting material has the advantage that production is carried out by natural fermentation processes. Thus, Perlastan glutamates such as SC 25 NKW, SL and SCG 50 ZPF fulfil the requirements for obtaining certifica[1]tion by various organisations such as COSMOS, ECOCERT, NATRUE, ECO-Label and Nordic Swan.
They can be readily used as the main components or secondary surfactants in formulations when certification as natural cosmetics is required. Their mild and particularly skin-friendly character is combined with a pronounced foaming behaviour and excellent aerobic and anaerobic biodegradability6 .
These positive features make the range of N-acyl sarcosinates and N-acyl glutamates surfactants of choice to meet increasingly sophisticated needs of consumers with respect to naturalness, health and performance.
In this article, the performance of amino acid-based surfactants and their formulations was examined by various methods with regard to their cleansing power, foam ability and foam structure and compared with conventional standard surfactants.
Both substances consist of the joining of a naturally occurring amino acid as a hydrophilic head to a long chain fatty acid as the hydrophobic tail. The ionic structure of the
amino acid part has a high affinity to proteinaceous surfaces such as skin and hair. Studies have shown that these amino acid-based surfactants form a protective monolayer on the skin and this prevents drying out of the skin. This protective monolayer also keeps more aggressive primary or secondary surfactants that may be present away from
the surface of the skin 1.
In stark contrast to standard surfactants such as SLS, SLES and cocamidopropyl betaine (CAPB), the N-acyl sarco[1]sinates and N-acyl glutamates show a capability to buf[1]fer the pH of formulations in the area of 5.5. This buffer capacity allows the preparation of stable formulations without any negative effects on the healthy condition of the skin 4 .
N-acyl glutamates such as Perlastan SC 25 NKW are well tolerated by the skin due to its low tendency to cause skin irritation (hypoallergenic) and absence of pore blocking (non-comedogenic) properties. The transepidermal water loss (TEWL) is a measure for the barrier stability of the epidermis and therefore an indicator for the irritant potential of substances.
In a clinical study the TEWL of human skin was investigated after application of two rinseoff formulations based upon sodium cocoyl glutamate and sodium lauroyl sarcosinate as main components. No significant change in skin moisture of the subjects could be determined, which is to be interpreted as a positive result. Furthermore, the tested rinse-off formulations showed a high degree of acceptance by the subjects in terms of their mild, nourishing, cleansing and foaming effect.
Glutamic acid as a starting material has the advantage that production is carried out by natural fermentation processes. Thus, Perlastan glutamates such as SC 25 NKW, SL and SCG 50 ZPF fulfil the requirements for obtaining certifica[1]tion by various organisations such as COSMOS, ECOCERT, NATRUE, ECO-Label and Nordic Swan.
They can be readily used as the main components or secondary surfactants in formulations when certification as natural cosmetics is required. Their mild and particularly skin-friendly character is combined with a pronounced foaming behaviour and excellent aerobic and anaerobic biodegradability6 .
These positive features make the range of N-acyl sarcosinates and N-acyl glutamates surfactants of choice to meet increasingly sophisticated needs of consumers with respect to naturalness, health and performance.
In this article, the performance of amino acid-based surfactants and their formulations was examined by various methods with regard to their cleansing power, foam ability and foam structure and compared with conventional standard surfactants.
 Figure 2: Static surface tension as a function of concentration. CMC of sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium lauroyl glutamate (Perlastan SL), sodium lauroyl sarcosinate (Perlastan L-30) and other common surfactants.
For this reason, it is common practice to include additional surfactant(s) with particularly low CMC in a formulation to reduce the CMC of the formulation as a whole.
A widely used and convenient formulation of cleansing products is the mixture of anionic surfactant SLES and amphoteric CAPB (CMC 0.12g/l). However, the strong ionic
structure of the sulphate group in SLES increases the potential for creating conditions that can show as skin irritation and can lead to intolerance especially if someone has
sensitive skin.
In this case the use of amino acid-based surfactants such as sodium cocoyl glutamate (CMC 0.4 g/l), sodium lauroyl glutamate (CMC 0.4 g/l) or sodium lauroyl
sarcosinate (CMC 0.4 g/l) can reduce the irritant effects and produce a milder formulation.
They may be added at very low levels for instance to SLES and amphoteric surfactants (betaines) formulations to produce significantly milder effects with positive influence on the health of the skin’s surface. The protective monolayer reduces adsorption of aggressive surfactants on the skin and at the same time loss of skin moisture is prevented.
Figure 2: Static surface tension as a function of concentration. CMC of sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium lauroyl glutamate (Perlastan SL), sodium lauroyl sarcosinate (Perlastan L-30) and other common surfactants.
For this reason, it is common practice to include additional surfactant(s) with particularly low CMC in a formulation to reduce the CMC of the formulation as a whole.
A widely used and convenient formulation of cleansing products is the mixture of anionic surfactant SLES and amphoteric CAPB (CMC 0.12g/l). However, the strong ionic
structure of the sulphate group in SLES increases the potential for creating conditions that can show as skin irritation and can lead to intolerance especially if someone has
sensitive skin.
In this case the use of amino acid-based surfactants such as sodium cocoyl glutamate (CMC 0.4 g/l), sodium lauroyl glutamate (CMC 0.4 g/l) or sodium lauroyl
sarcosinate (CMC 0.4 g/l) can reduce the irritant effects and produce a milder formulation.
They may be added at very low levels for instance to SLES and amphoteric surfactants (betaines) formulations to produce significantly milder effects with positive influence on the health of the skin’s surface. The protective monolayer reduces adsorption of aggressive surfactants on the skin and at the same time loss of skin moisture is prevented.
 Figure 3: Reduction of the critical micelle concentration (CMC) of sodium cocoyl glutamate (PerlastanSC 25 NKW) and sodium lauroyl sarcosinate (Perlastan L-30) by equivalent combination with coco glucoside (1:1).
When the surfactants are mixed with non-ionic surfactant coco glucoside (CMC 0.045 g/l) in equivalent ratio, the total-CMC is reduced from 0.4 g/l to 0.12g/l or 0.2 g/l (Fig 3). This means that a lower concentration of surfactant is needed to obtain same cleansing performance with excellent foam ability and mildness of the product.
Figure 3: Reduction of the critical micelle concentration (CMC) of sodium cocoyl glutamate (PerlastanSC 25 NKW) and sodium lauroyl sarcosinate (Perlastan L-30) by equivalent combination with coco glucoside (1:1).
When the surfactants are mixed with non-ionic surfactant coco glucoside (CMC 0.045 g/l) in equivalent ratio, the total-CMC is reduced from 0.4 g/l to 0.12g/l or 0.2 g/l (Fig 3). This means that a lower concentration of surfactant is needed to obtain same cleansing performance with excellent foam ability and mildness of the product.
 Figure 4: Ross-Miles test with sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium lauroylsarcosinate (Perlastan L-30) and other surfactants (active content of 3 g/l).
This test was performed to determine foam ability and foam stability of surfactant solutions with an active content of 3 g/l (above the CMC). A defined volume of foam was generated by a foam dispenser and the foam height was measured as a function of time. The developed foam height of Perlastan surfactants is in a general equivalent to high foaming standard surfactants such as sulphates and betaines.
It is a positive additional effect that these surfactants form foams that are extremely stable and microporous in structure. In an additional test where defined foam was formed by means of a foam dispenser, the amount of liquid draining from the foams was measured on a time dependent basis.
This is an indication of the amount of liquid in the foam. The results show that sarcosinates and more particularly glutamates provide a stable foam with a high water content that is stable over a longer period (Figure 5).
Figure 4: Ross-Miles test with sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium lauroylsarcosinate (Perlastan L-30) and other surfactants (active content of 3 g/l).
This test was performed to determine foam ability and foam stability of surfactant solutions with an active content of 3 g/l (above the CMC). A defined volume of foam was generated by a foam dispenser and the foam height was measured as a function of time. The developed foam height of Perlastan surfactants is in a general equivalent to high foaming standard surfactants such as sulphates and betaines.
It is a positive additional effect that these surfactants form foams that are extremely stable and microporous in structure. In an additional test where defined foam was formed by means of a foam dispenser, the amount of liquid draining from the foams was measured on a time dependent basis.
This is an indication of the amount of liquid in the foam. The results show that sarcosinates and more particularly glutamates provide a stable foam with a high water content that is stable over a longer period (Figure 5).
 Figure 5: Foam Stability: Time-dependent drainage amount of sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium cocoyl glutamate/coco glucoside/xanthan gum, sodium lauroyl sarcosinate (Perlastan L-30) and other surfactants (active content of 3 g/l).
CAPB and coco glucoside provided the fastest water loss and thereby may be described as being the most unstable and driest foam. A wet foam consistency as demonstrated by amino acid-based surfactants allows a particularly easy application of the product on skin and leaves a pleasant skin-feel.
It was also demonstrated that the relatively unstable foam of a 0.3% coco glucoside solution (active content of 3 g/l) was noticeably stabilised by an equivalent amount of sodium cocoyl glutamate (1:1) and xanthan gum (1%) and gave the best stability under these test conditions.
Figure 5: Foam Stability: Time-dependent drainage amount of sodium cocoyl glutamate (Perlastan SC 25 NKW), sodium cocoyl glutamate/coco glucoside/xanthan gum, sodium lauroyl sarcosinate (Perlastan L-30) and other surfactants (active content of 3 g/l).
CAPB and coco glucoside provided the fastest water loss and thereby may be described as being the most unstable and driest foam. A wet foam consistency as demonstrated by amino acid-based surfactants allows a particularly easy application of the product on skin and leaves a pleasant skin-feel.
It was also demonstrated that the relatively unstable foam of a 0.3% coco glucoside solution (active content of 3 g/l) was noticeably stabilised by an equivalent amount of sodium cocoyl glutamate (1:1) and xanthan gum (1%) and gave the best stability under these test conditions.
